The MEVION S250-FIT™ creates a new pathway for European cancer centers to integrate proton therapy into existing radiotherapy programs
Proton therapy is entering the LINAC vault. At ESTRO 2026, Mevion Medical Systems will introduce the MEVION S250-FIT Proton Therapy System to the European radiation oncology community, the first proton therapy system designed for installation in a standard radiation therapy vault.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260507172087/en/
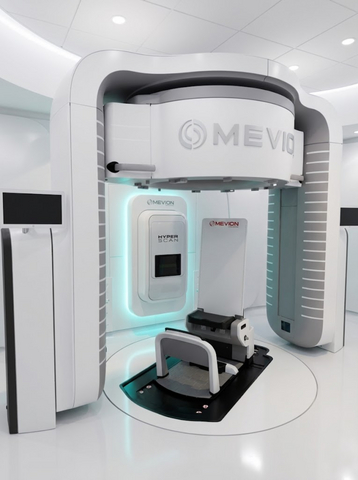
The MEVION S250-FIT™ Proton Therapy System
Now both U.S. FDA-cleared and CE-marked under Regulation (EU) 2017/745, the S250-FIT creates a new pathway for cancer centers to bring proton therapy into existing LINAC-based treatment environments, aligning advanced proton capability with the infrastructure, workflows, and capital planning of modern radiation oncology.
Stanford Medicine: First S250-FIT Installation
On April 7, 2026, Stanford Medicine unveiled the world’s first S250-FIT installation, developed in collaboration with Leo Cancer Care. The system was fully installed within a standard 110 m² (1,200 sq ft) LINAC vault inside the existing Stanford Medicine Cancer Center in Palo Alto, without constructing a new building.
“With FIT™, proton therapy is no longer limited by infrastructure,” said Tina Yu, Ph.D., CEO and President of Mevion Medical Systems. “For the first time, it can be deployed within the same clinical and operational framework as conventional radiotherapy. For European health systems that have long recognized the benefits of proton therapy but faced significant infrastructure and financial barriers, this changes the question from whether to build a separate proton facility to how to integrate proton therapy into the radiotherapy programs they already operate.”
Advanced Capability in a Compact Footprint
The S250-FIT delivers Intensity Modulated Proton Therapy via Mevion's HYPERSCAN® pencil beam scanning with the Adaptive Aperture® proton multi-leaf collimator and is designed to support DirectARC™ proton arc therapy. The system supports advanced image-guided and adaptive workflows and is FLASH research-ready.* The system is paired with Leo Cancer Care’s Marie® Upright Patient Positioning and CT Imaging System, the first commercial upright treatment platform with an integrated diagnostic CT, offering potential benefits in patient comfort and supporting organ motion management during treatment.
Built on Decades of U.S. and European Clinical Experience
The S250-FIT shares its core technology lineage with the MEVION S250i®, in clinical operation at Siteman Cancer Center at Washington University School of Medicine in the U.S. and at ZON-PTC at Maastro Clinic in the Netherlands, giving Mevion more than a decade of combined U.S. and European clinical experience with compact, single-room proton therapy. Mevion has now signed contracts with nine leading institutions globally for the S250-FIT, including Stanford Health Care, Loma Linda University Health, UNC Health, BayCare Health System, Atlantic Health System, and University of Nebraska Medical Center.
Meet Mevion at ESTRO 2026
Visit Mevion at Booth #C08:89 to engage with the Mevion team and discuss site planning, clinical workflows, and integration pathways with our clinical and engineering leadership.
*The FLASH Research Kit is not currently available for commercial sale or for human clinical use.
About Mevion Medical Systems
Mevion Medical Systems is the leading provider of compact proton therapy systems for cancer care. Dedicated to advancing the design and accessibility of proton therapy worldwide, Mevion pioneered the single-room platform and continues to further the science and application of proton therapy. Since 2013, Mevion’s compact proton therapy single-room systems have been used by leading cancer centers for treating patients. Mevion’s series of products, including the flagship MEVION S250i and MEVION S250-FIT™ with HYPERSCAN pencil beam scanning, represent the world’s most compact proton therapy systems that eliminate the obstacles of size, complexity, and cost. Mevion is headquartered in Littleton, Massachusetts, with a presence in Europe and Asia. For more information, please visit www.mevion.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260507172087/en/
Contacts
Media Contact:
Jacqueline Abner-Pongratz
Jacqueline.Pongratz@mevion.com





